Gujarat Corona Update: 15/8/20 Official Press Note
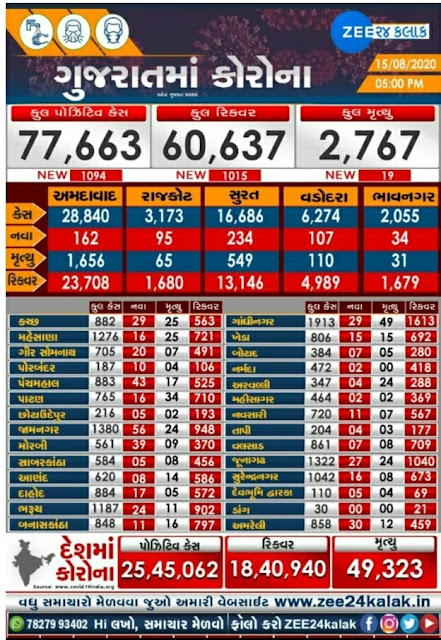
Gujarat Corona Update: 1015 new patients, 1094 patients recovered, 19 deaths
Corona's condition in the state is getting worse day by day. Today 1015 patients were registered. On the other hand, 1094 patients have recovered and returned home. The government is also claiming that the number of tests in the state is increasing day by day. A total of 51217 tests were conducted in the state today. Which is 787.95 per million per day according to the population of the state.
A total of 12,62,264 tests have been conducted in the state so far. A total of 1090 cases have been registered from various districts of the state and 049 from other states. Today, 1,015 patients have recovered and gone home.
A total of 5,01,746 persons have been quarantined in different districts of the state to date. Out of which 5,00,875 persons have been home quarantined. While 921 persons are being kept in facility quarantine.
If we talk about active patients, there are currently 14359 active cases in the state. 76 on the ventilator. While 14283 people are stable. 60537 people have been discharged. 2767 people have been killed. Today, 19 people have been killed. A total of 19 people, including 5 in Surat, 4 in Surat Corporation, 3 in Ahmedabad Corporation, 2 in Vadodara Corporation, 1 in Gir Somnath, 1 in Junagadh, 1 in Mehsana, 1 in Morbi and 1 in Rajkot, have been killed.
Image source
Gujarat Corona Update: 1015 new patients, 1094 patients recovered, 19 deaths
Corona's condition in the state is getting worse day by day. Today 1015 patients were registered. On the other hand, 1094 patients have recovered and returned home. The government is also claiming that the number of tests in the state is increasing day by day. A total of 51217 tests were conducted in the state today. Which is 787.95 per million per day according to the population of the state.
A total of 12,62,264 tests have been conducted in the state so far. A total of 1090 cases have been registered from various districts of the state and 049 from other states. Today, 1,015 patients have recovered and gone home.
A total of 5,01,746 persons have been quarantined in different districts of the state to date. Out of which 5,00,875 persons have been home quarantined. While 921 persons are being kept in facility quarantine.
If we talk about active patients, there are currently 14359 active cases in the state. 76 on the ventilator. While 14283 people are stable. 60537 people have been discharged. 2767 people have been killed. Today, 19 people have been killed. A total of 19 people, including 5 in Surat, 4 in Surat Corporation, 3 in Ahmedabad Corporation, 2 in Vadodara Corporation, 1 in Gir Somnath, 1 in Junagadh, 1 in Mehsana, 1 in Morbi and 1 in Rajkot, have been killed.
 |
| Gujarat Corona Update: 15/8/20 Official Press Note |
Official press note
date 15/8/20 Corona Official Press note Read Click Here
COVAXIN: The good news is that the native corona vaccine has arrived
The first phase of the clinical trial of the Covid-19 vaccine being developed by Bharat Biotech-ICMR has been successful. According to an ET report, after the initial results of the trial, it is said that the vaccine is completely safe. Human trials of India Biotech and Zydus Cadillac vaccine are underway in 6 cities. Coronavirus vaccine tested on 375 volunteers in 12 cities in India.
The first phase of the clinical trial of the Covid-19 vaccine being developed by Bharat Biotech-ICMR has been successful. According to an ET report, after the initial results of the trial, it is said that the vaccine is completely safe. Human trials of India Biotech and Zydus Cadillac vaccine are underway in 6 cities. Coronavirus vaccine is being tested on 375 volunteers in 12 centers in India. Each volunteer is being given two doses of the vaccine and is also being monitored.
The vaccine did not cause any side effects
Savita Verma, the team leader of the ongoing trial at PGI Rohtak, said the vaccine was safe. None of the volunteers we have vaccinated with have had the opposite effect. Volunteers are now being prepared for a second dose. This is previously being done by the blood sample collector of the investigating volunteers. Blood samples will be used to test the immunogenicity of the vaccine.
The second phase of blood sample collection started
"We now know that the vaccine is completely safe," said Savita Verma. Now we will know in the second stage how effective the vaccine is. For this, we have started collecting blood samples from people.
16 volunteers at the AIIMS were vaccinated
Sanjay Rai, the principal investigator at the All India Institute of Medical Sciences (AIIMS) in Delhi, said the vaccine was safe. The AIIMS recruited 16 volunteers for the India Biotech Vaccine test.
The vaccine is being developed in collaboration with ICMR
India is also involved in the race to develop a safe coronavirus vaccine. The government itself is monitoring the whole process. Covin is India's first vaccine and has been developed by Bharat Biotech in collaboration with ICMR.
The company will now approach the Drug Controller General of India in the second phase once the expected security results are obtained from all the 12 locations. Another investigator, speaking on condition of anonymity, said the vaccine would be available in the first half of next year if all goes well.
COVAXIN: The good news is that the native corona vaccine has arrived
The first phase of the clinical trial of the Covid-19 vaccine being developed by Bharat Biotech-ICMR has been successful. According to an ET report, after the initial results of the trial, it is said that the vaccine is completely safe. Human trials of India Biotech and Zydus Cadillac vaccine are underway in 6 cities. Coronavirus vaccine tested on 375 volunteers in 12 cities in India.
The first phase of the clinical trial of the Covid-19 vaccine being developed by Bharat Biotech-ICMR has been successful. According to an ET report, after the initial results of the trial, it is said that the vaccine is completely safe. Human trials of India Biotech and Zydus Cadillac vaccine are underway in 6 cities. Coronavirus vaccine is being tested on 375 volunteers in 12 centers in India. Each volunteer is being given two doses of the vaccine and is also being monitored.
The vaccine did not cause any side effects
Savita Verma, the team leader of the ongoing trial at PGI Rohtak, said the vaccine was safe. None of the volunteers we have vaccinated with have had the opposite effect. Volunteers are now being prepared for a second dose. This is previously being done by the blood sample collector of the investigating volunteers. Blood samples will be used to test the immunogenicity of the vaccine.
The second phase of blood sample collection started
"We now know that the vaccine is completely safe," said Savita Verma. Now we will know in the second stage how effective the vaccine is. For this, we have started collecting blood samples from people.
16 volunteers at the AIIMS were vaccinated
Sanjay Rai, the principal investigator at the All India Institute of Medical Sciences (AIIMS) in Delhi, said the vaccine was safe. The AIIMS recruited 16 volunteers for the India Biotech Vaccine test.
The vaccine is being developed in collaboration with ICMR
India is also involved in the race to develop a safe coronavirus vaccine. The government itself is monitoring the whole process. Covin is India's first vaccine and has been developed by Bharat Biotech in collaboration with ICMR.
The company will now approach the Drug Controller General of India in the second phase once the expected security results are obtained from all the 12 locations. Another investigator, speaking on condition of anonymity, said the vaccine would be available in the first half of next year if all goes well.








No comments:
Post a Comment